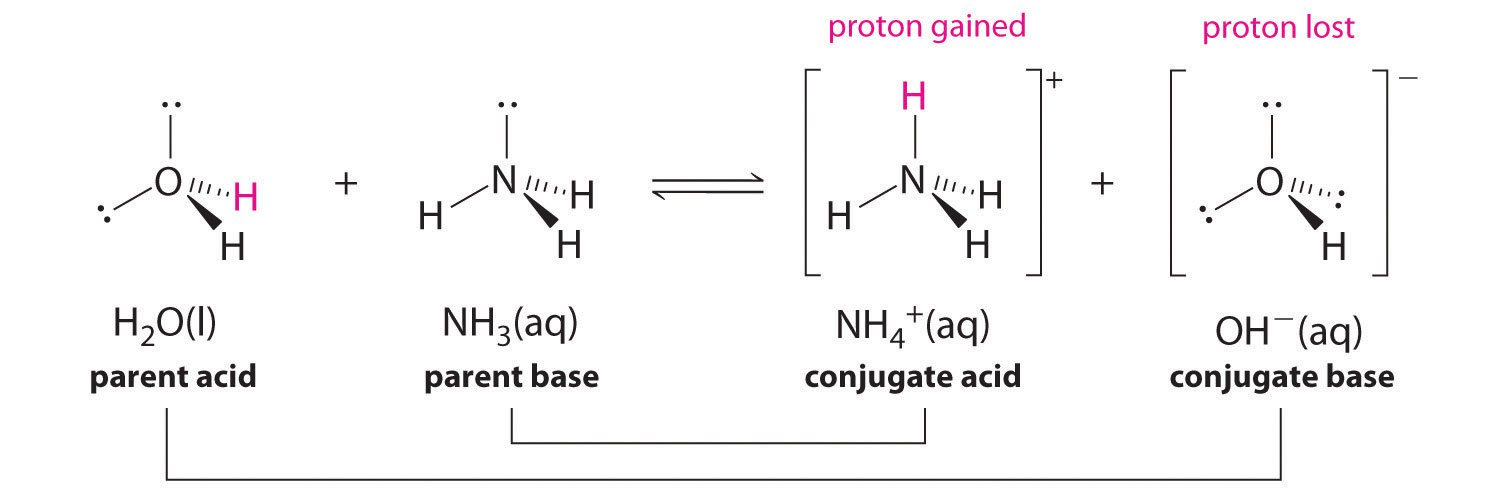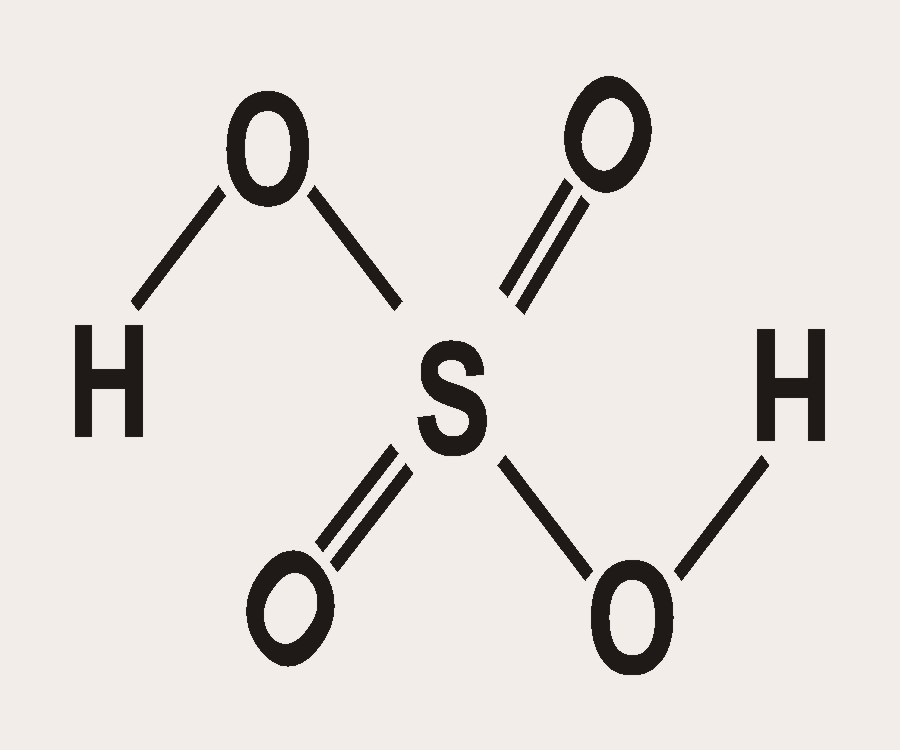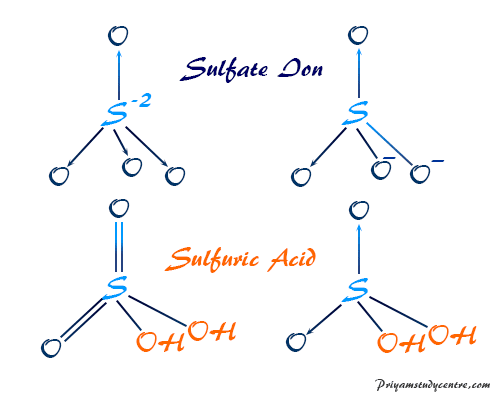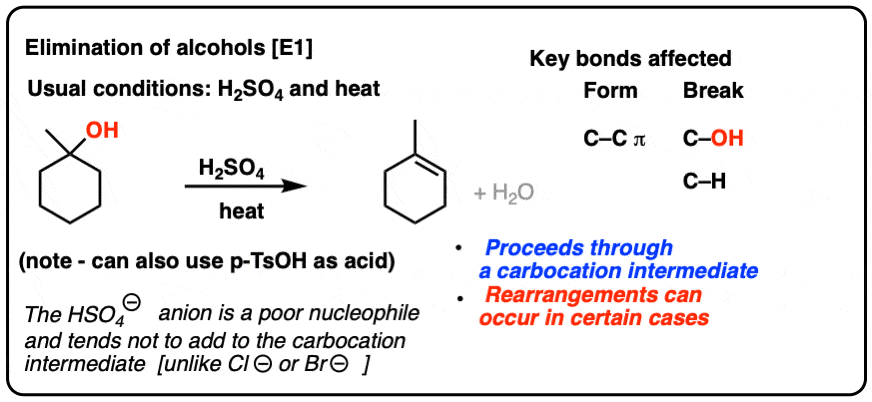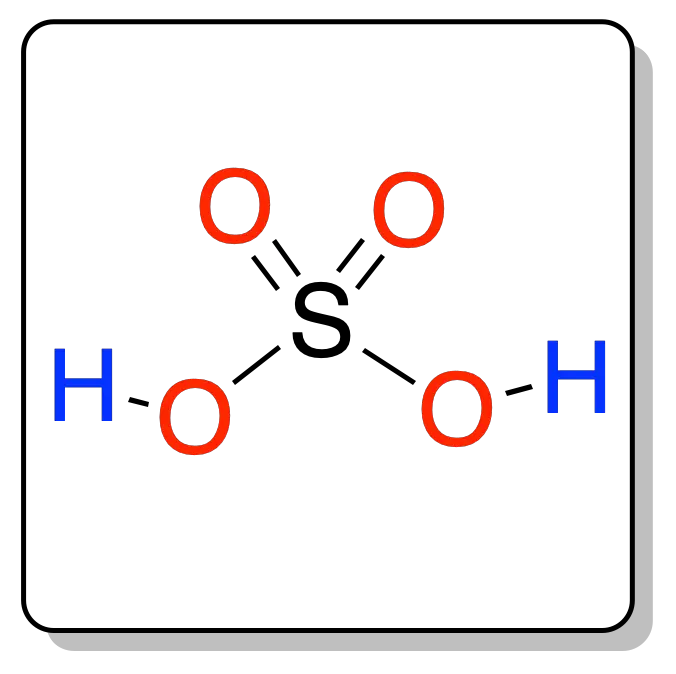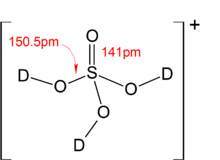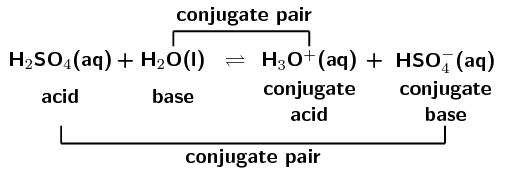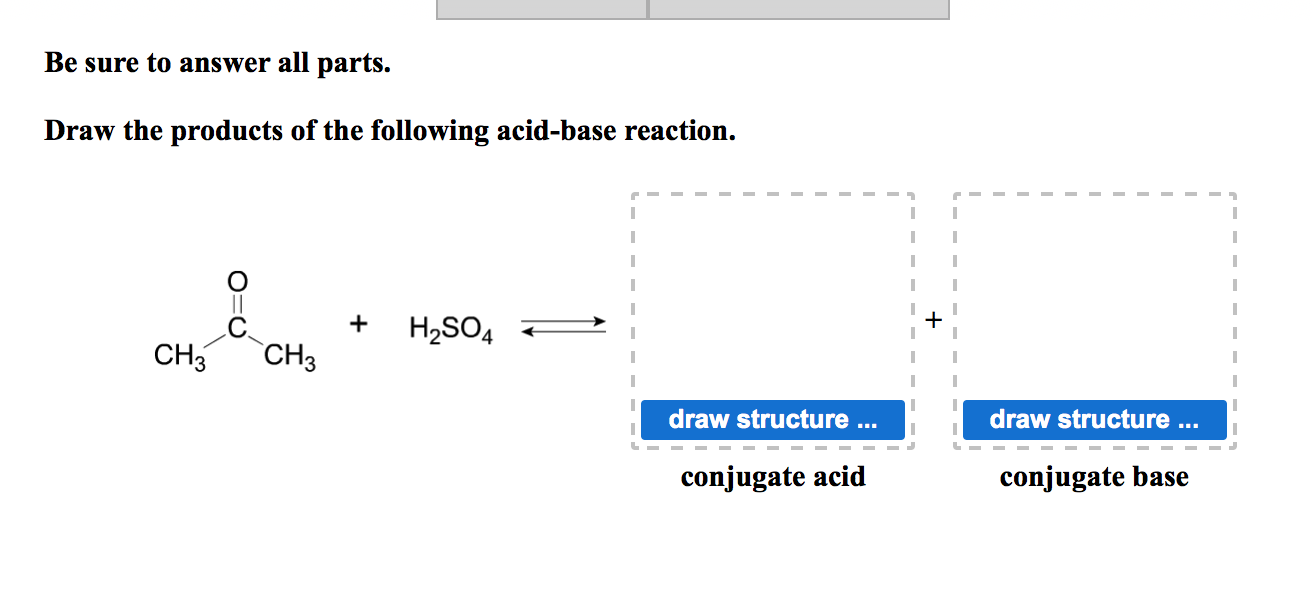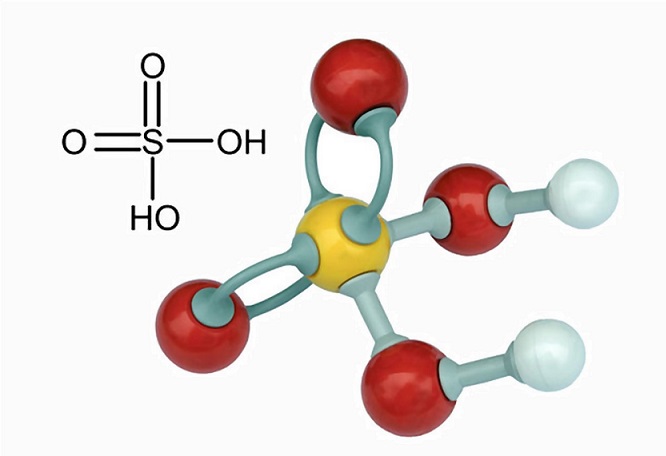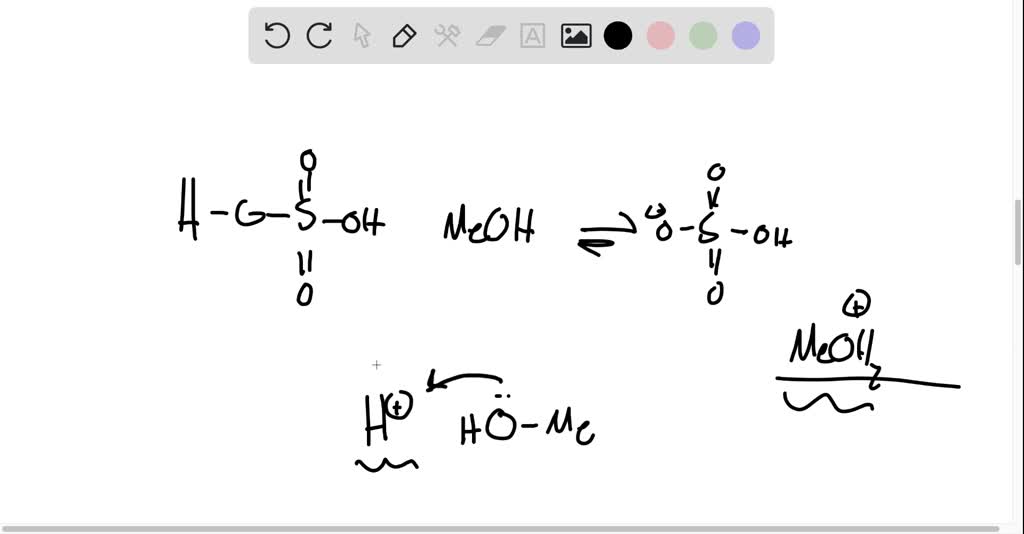
SOLVED: Organic Chemistry Write the products of the following acid-base reactions: (a) CH3OH + H2SO4 <–> ? (b) CH3OH + NaNH2 <–> ? (c) CH3NH3 + Cl- + NaOH <–> ? -

SOLVED: Identify each as an Acid, Base or Salt then write the equation or reaction that shows how each behaves in water: A) H2SO4 B) NaOH C CaClz D) Mg(OH)2 E) NH3